Nature publishes trial results of β-thalassaemia gene therapy
Nature published results of a phase 1 trial using CS-101 for β-thalassaemia treatment. The trial demonstrated early and lasting transfusion independence in patients. This development comes as gene editing technologies gain momentum.
Trial Results
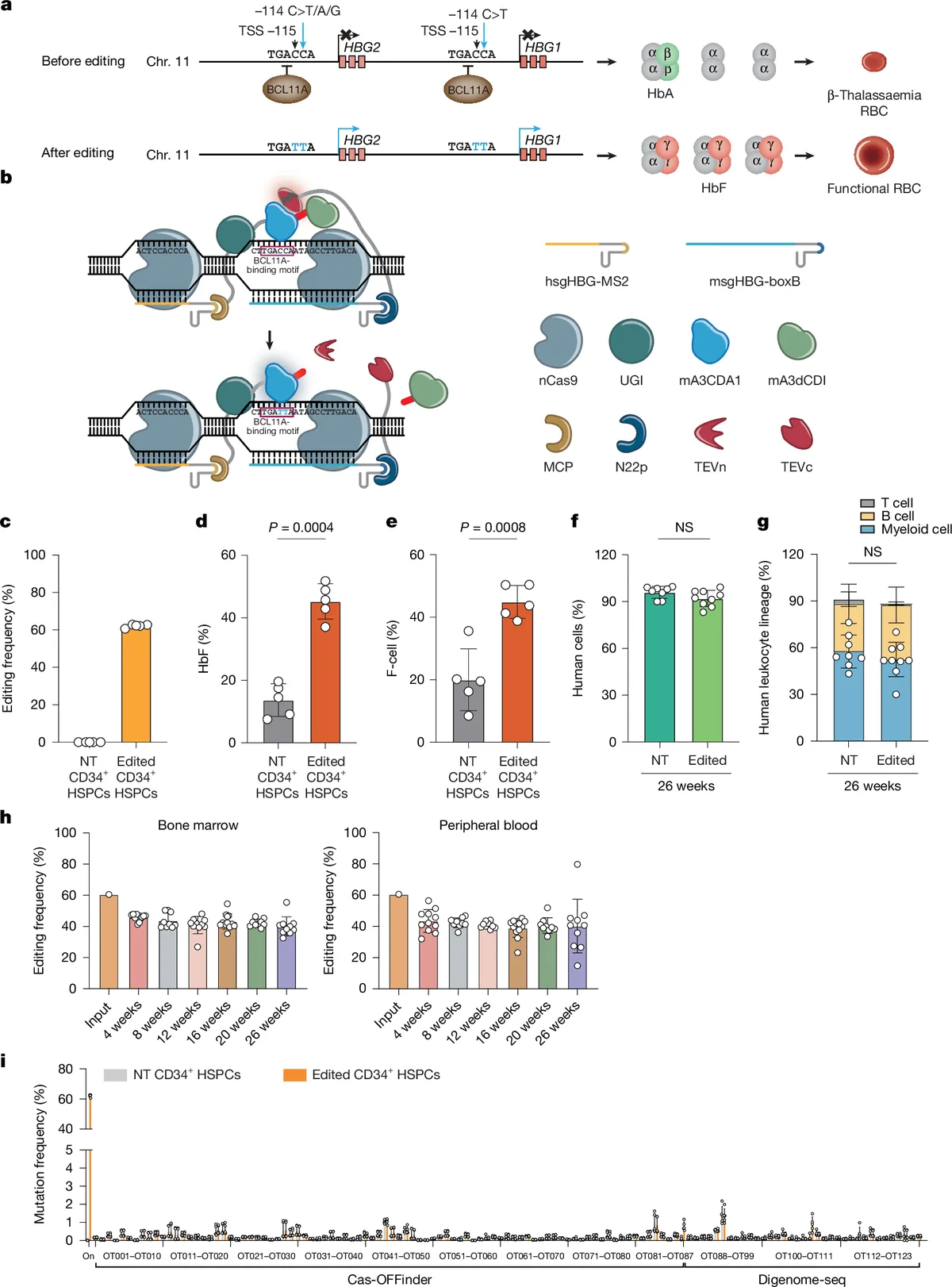
The phase 1 trial involved a single infusion of CS-101, which uses a transformer base editor to modify CD34+ cells. The treatment aimed to reactivate fetal haemoglobin production in patients with β-thalassaemia. Conducted by a team of researchers, the trial showed promising results with patients achieving transfusion independence. The study was published in Nature on April 8, 2026.
Gene Editing Technologies
The CS-101 therapy represents a significant advancement in gene editing, particularly for blood disorders. The transformer base editor used in the trial is part of a new wave of genetic technologies. These technologies are being developed by institutions such as CRISPR Therapeutics and Editas Medicine. The trial's success may encourage further investment and research in the field.
What's Next
Further clinical trials are expected to assess the long-term safety and efficacy of CS-101. It remains unclear how quickly this therapy could become widely available to patients.
2 sources
Nature publishes trial results of β-thalassaemia gene therapy